Project & Team
Clinical Project Description of CMT-MOD Biomarker Study
Recruitment
Patient Recruitment only via E-Mail:
contact@cmt-mod.org
Sponsor
This study is sponsored by the AFM TÉLÉTHON.
Study Title
Full Title: A multi-omic approach to the identification of novel biomarkers in early Charcot-Marie-Tooth 1A disease (CMT1A) (CMT-MODs)
Clinical Phase
This is a biomarker study, not associated with a specific clinical phase.
Coordinators
Lead Coordinator:
Univ.-Prof. Dr. med. Michael W. Sereda
University Medical Centre Göttingen (UMG), Department of Neurology
Address: Robert-Koch-Str. 40, 37075 Göttingen, Germany
Email: mwsereda@med.uni-goettingen.de or sereda@mpinat.mpg.de
Co-Coordinator:
Prof. Shahram Attarian, M.D., Ph.D.
Neuromuscular Disease and ALS Reference Center, Timone University Hospital, Aix-Marseille University
Address: CHU Timone, 264 rue Saint Pierre, 13385 Marseille Cedex 05, France
Email: cmt1a@ap-hm.fr
Participating Centers
The project involves multiple research centers led by principal investigators, including:
University Medical Centre Göttingen (UMG), Germany
Principal Investigator: Univ.-Prof. Dr. Michael Sereda
Timone University Hospital, Aix-Marseille University, France
Principal Investigator: Prof. Shahram Attarian

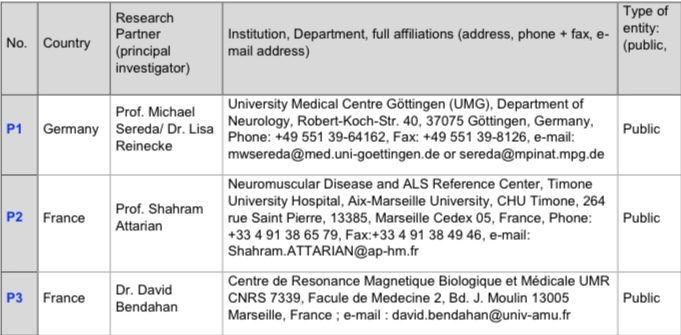
Research Partners and Collaborators
Research Partners (Direct Funding)
These Institutions are directly involved in the project´s core activities and receive funding to support their work.
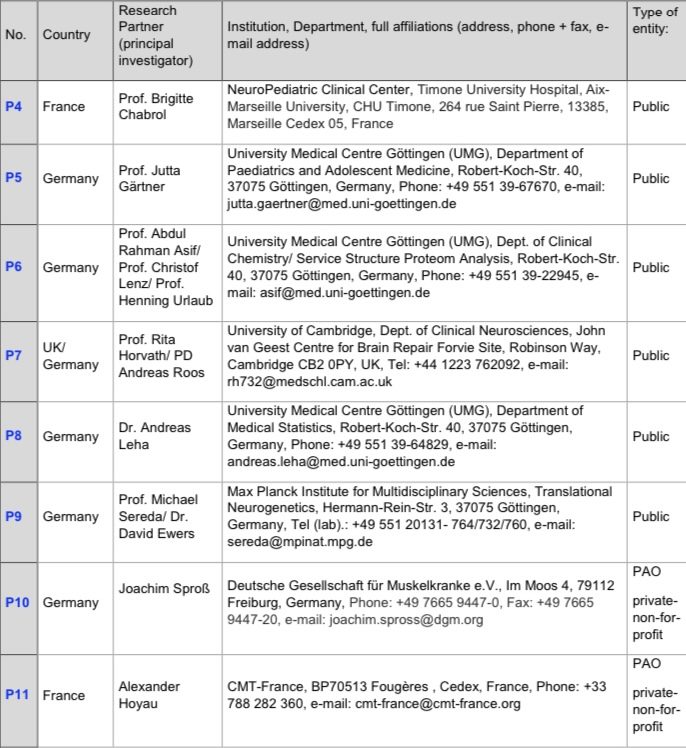
Collaborating Partners (Indirect funding via P1 or P2)
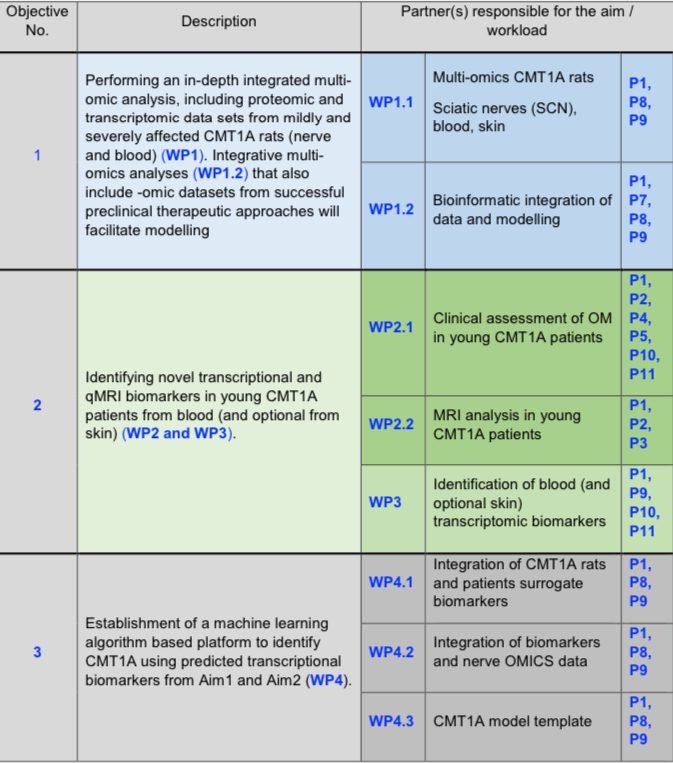
Objectives (primary and secondary)
Estimated Study Duration
Inclusion Period: 6 months
Study Duration: 12 months
Total Timeframe: 18 months (from first patient enrolled to last patient completed).
